 Once we determine the energy for the last step, we reverse the equation and change the sign for the lattice energy. The lattice energy is the reverse of the formation of the solid from its ions. The net energy change for the reaction is -1228 kJ/mol. 
Calculate the lattice energy for CaF 2.Įnergy of formation for one mole of CaF 2 from its elements = -1228 kJ/mol The lattice energy for SrCl 2 is equal to +2123 kJ.Įxercise 5. Subtract the energies from -803.7 kJ and solve for the formation of the solid from its ions–this will be x. We know the net energy change is -803.7 kJ/mol. The net energy change for the reaction is -803.7 kJ/mol. 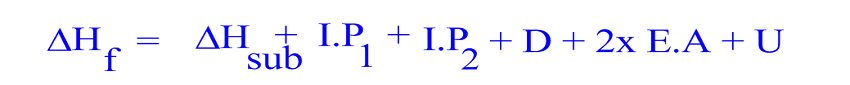 
The sublimation energy for Sr is 164 kJ/mol, E i1 = +549.5 kJ/mol, E i2 = +1064.2 kJ/mol, E ea for Cl (g) = -348.6 kJ/mol, energy for the formation of one mole of SrCl 2 from its elements = -803.7 kJ/mol, and the bond dissociation energy for Cl 2 (g) = +243 kJ/mol. Which has the highest lattice energy, Na 3P or Na 2S?Įxercise 4. Order the following ionic compounds from lowest to highest lattice energy. The lattice energy is the reverse of Step 6.Įxercise 2. Consider the following Born-Haber cycle for MgF 2. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
